If you received a Cartiva toe implant and are now dealing with ongoing pain, loss of motion, implant failure, or revision surgery, you may have the right to pursue a Cartiva lawsuit against the manufacturer. Patients across the country allege that the Cartiva Synthetic Cartilage Implant failed to perform as promised and instead left them with worsening symptoms, additional procedures, and significant disruption to their daily lives. Wilentz, Goldman & Spitzer, P.A. is actively litigating Cartiva implant claims for individuals who were harmed after receiving this device.
The Cartiva implant was marketed as an alternative to fusion for arthritis in the big toe joint. For many patients, the appeal was obvious: preserve motion, reduce pain, and avoid a more invasive outcome. But for a growing number of people, that is not what happened. Instead of lasting relief, they experienced persistent pain, stiffness, subsidence, implant shrinkage, bone loss, and the need for revision surgery, often ending in the same fusion procedure they hoped to avoid in the first place. The FDA now lists a Class II recall for the Cartiva SCI covering all lots distributed from July 2016 through October 2024, and federal Cartiva cases have now been centralized into MDL No. 3172 in the Eastern District of Arkansas.
If you are suffering after a Cartiva implant procedure, you are not alone, and your symptoms should not be dismissed as simply part of the normal healing process. Our firm is prepared to evaluate whether your injuries may support a claim and whether you may be entitled to compensation for revision surgery, additional medical care, lost income, pain and suffering, and other losses.
What is the Cartiva lawsuit about?
Cartiva lawsuits allege that the Cartiva Synthetic Cartilage Implant was defectively designed and/or sold without adequate warnings, resulting in premature failure and serious complications.
Has the Cartiva implant been recalled?
Yes. The FDA lists a Class II recall affecting all lots distributed from July 2016 to October 2024.
Is there a Cartiva MDL?
Yes. Federal Cartiva cases were centralized into MDL No. 3172 on February 5, 2026 in the U.S. District Court for the Eastern District of Arkansas.
Who is the lawsuit against?
These claims are against the device manufacturer, not your doctor.
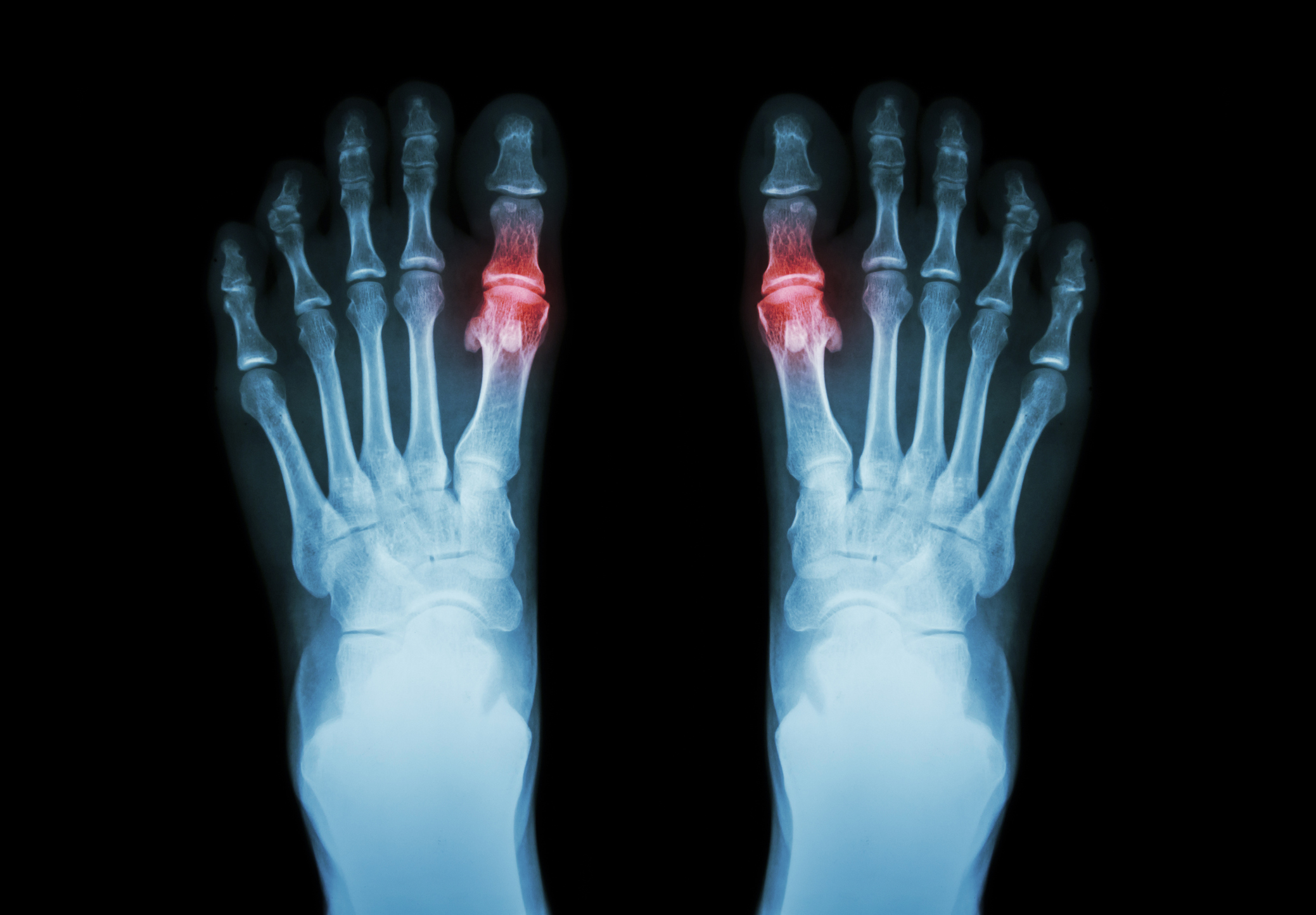
The Cartiva Synthetic Cartilage Implant, often called the Cartiva SCI, is a synthetic hydrogel implant used to treat arthritis in the first metatarsophalangeal joint, which is the joint at the base of the big toe. It was promoted as a motion-preserving alternative to fusion for patients suffering from hallux rigidus, a condition that can cause chronic pain, stiffness, and difficulty walking.
For many patients, that promise was central to the treatment decision. Fusion can be an effective procedure, but it eliminates motion in the toe joint. The Cartiva Implant was presented as a way to relieve pain while preserving movement. When the implant fails, however, patients may end up facing exactly what they tried to avoid: more pain, more surgery, and, in many cases, conversion to fusion with a more complicated recovery.
Patients are filing lawsuits because they allege that the Cartiva implant failed at unacceptable rates and caused serious complications that required additional treatment. These claims generally center on allegations that the device was defective and that patients and doctors were not adequately warned about the true risks of failure.
A failed Cartiva implant can affect nearly every part of a person’s daily routine. Walking, exercising, climbing stairs, standing for work, and even wearing certain shoes can become difficult or painful. Some patients report that they were worse off after the implant than they were before surgery. Others required revision procedures, hardware placement, bone grafting, or fusion after the implant did not hold up as expected. The recurring pattern across these cases is that people sought relief and motion preservation, but instead were left with further injury and loss.
Cartiva implant failure symptoms and complications
Common symptoms and complications associated with Cartiva implant failure may include:
Some patients are told their recovery is simply taking longer than expected. But if you are months or years out from surgery and still experiencing pain, stiffness, deformity, or loss of function, that should be taken seriously. If your implant is failing, the symptoms will only worsen.
When a Cartiva implant fails, the consequences can be much more significant than simply undergoing a second procedure. Because bone is removed when the implant is placed, taking the implant out does not necessarily restore the joint to its prior condition. Many patients ultimately require a fusion procedure after failure, sometimes with bone grafting and hardware to address the remaining defect. That can make the revision more invasive than a primary fusion would have been in the first place.
This matters not only medically, but legally. A failed revision or a complicated fusion after Cartiva may increase the extent of the damages in a case because the patient may face additional surgeries, more pain, reduced mobility, longer recovery, and more significant lifestyle limitations.
The FDA lists a Class II recall for the Cartiva Synthetic Cartilage Implant. The recall record identifies all lots distributed from July 2016 to October 2024 and includes multiple implant sizes. The underlying safety notice said the purpose was to provide updated post-market safety data regarding a higher-than-expected occurrence of device removal, revision, and conversion to fusion.
A recall does not automatically mean every patient has a lawsuit, but it is highly relevant evidence in evaluating these claims. It may support the broader allegation that the device did not perform as represented and that the risks were more serious than many patients were led to believe. If you received a Cartiva implant during the affected distribution period and later experienced complications, that timing may be important to your legal evaluation.
On February 5, 2026, the Judicial Panel on Multidistrict Litigation centralized federal Cartiva lawsuits into MDL No. 3172, titled In re: Cartiva Synthetic Cartilage Implant Products Liability Litigation, in the Eastern District of Arkansas.
An MDL is not the same thing as a class action. In a class action, one case proceeds on behalf of a larger group. In an MDL, each person still maintains an individual case, but pretrial proceedings such as discovery, document production, and common expert issues are coordinated before one judge. That structure is often used in complex product liability litigation because it promotes efficiency while preserving individual claims.
For injured patients, the MDL can be significant because it creates a centralized federal process for developing evidence against the manufacturer. It also means that the litigation is advancing in a more formal and organized way than before.
You may be a candidate for a Cartiva lawsuit evaluation if:
Every case is different, but compensation in a Cartiva implant lawsuit may include damages for:
The value of a claim will depend on the severity of the complications, whether revision surgery was required, the long-term effect on your mobility, and the strength of the supporting medical evidence.
Your first priority should be your health. If you are still suffering after a Cartiva implant, seek medical evaluation from your treating physician or another qualified orthopedic or foot-and-ankle specialist. You may need imaging, follow-up assessment, and a treatment plan to determine whether the implant has failed and what options are available.
After addressing your immediate medical needs, preserve as much documentation as possible. That can include operative records, imaging, follow-up notes, billing records, and any recommendation for revision surgery or fusion. In device cases, the medical evidence matters. The sooner your records are organized and reviewed, the better positioned you are to understand whether you may have a viable claim.
Now that the MDL is the primary path for these claims, your choice of attorney is more important than ever. You need a firm with the experience to navigate complex mass torts and a fee structure that respects your recovery. So why choose Wilentz, Goldman & Spitzer, P.A.?
As the litigation moves forward in the MDL, Wilentz offers a combination of experience, local insight, and a commitment to fair pricing.
Fairer Fees: Save Money on Your Recovery
Our fee structure is designed to put more money into your pocket. By choosing the Wilentz firm, you cap your contingency fee at 33 1/3%. Firms handling MDL cases often charge their clients 40% or more, meaning you could pay thousands—or even tens of thousands—of dollars more in fees for the exact same recovery. Our firm can also offer you an hourly billing retention if you prefer not to agree to a contingency fee arrangement.
|
Firm/Venue |
Contingency Fee |
Fee Increase |
|
Wilentz, Goldman & Spitzer, P.A. |
33 1/3% |
— |
|
Out-of-State Counsel (MDL) |
40% or more |
You pay 6.67% or more in extra fees |
25+ Years of Proven Experience
Experience in medical device litigation is not all the same. Our team has over 25 years of experience holding medical device manufacturers accountable. We understand the science, the regulatory history of the Cartiva implant, and the tactics used by large corporations to defend these products.
Deep Knowledge of the Manufacturer
Stryker Corporation, the parent company of Cartiva, has its Orthopaedics division headquartered right here in New Jersey. Our firm has successfully litigated against Stryker for decades. We have an intimate understanding of their corporate structure and a long history of achieving multi-million dollar recoveries for victims of their defective medical devices. Please note that results achieved in prior matters are not meant to be a guarantee of success as the facts and legal circumstances vary from matter to matter.
Don't Get Lost in the Shuffle
In an MDL with hundreds or thousands of plaintiffs, it is easy for an individual’s story to be sidelined. At Wilentz, we provide the resources of a large, established firm with the personalized attention of a dedicated legal team. We make sure your specific injuries and complications are documented and prioritized.
Attorney Joshua S. Kincannon and the Mass Tort team at Wilentz are ready to represent you in the Cartiva MDL. We offer the experience you need to go up against a global corporation and a fee structure that prioritizes your financial future.
Results achieved in prior matters are not meant to be a guarantee of success as the facts and legal circumstances vary from matter to matter.
If you are living with pain, stiffness, loss of motion, or revision surgery after receiving a Cartiva toe implant, you should not assume this is just how the procedure was supposed to end. You have legal rights, and your case may deserve review in light of the recall and the growing federal litigation.
To speak with an attorney about your legal options, call 732-313-2323 for a free consultation.
Navigating the complexities of medical device litigation can be overwhelming. We've compiled answers to some common questions to help you understand your options if you've been affected by a defective Cartiva implant.
No. Federal Cartiva litigation is proceeding through an MDL, not a class action. That means each person keeps an individual claim, even while certain pretrial proceedings are coordinated.
No. These claims are focused on the manufacturer and the alleged defects or warnings associated with the device.
Yes. Every state has statutes of limitations that can affect how long you have to file. The applicable deadline can depend on where you live and when you knew or reasonably should have known that the implant may have caused your injuries. That makes prompt legal review important.
Wilentz handles these cases on a contingency fee basis, meaning there are no upfront attorneys’ fees and the firm is paid only if there is a recovery.
Joshua S. Kincannon
Shareholder
Lynne M. Kizis
Co-Chair, Mass Tort/Class Action Team
Shareholder